Introduction
The Japan Liquid Biopsy Cancer Screening Market is emerging as a critical segment within the global oncology diagnostics landscape, driven by technological innovation, rising cancer prevalence, and Japan’s strong healthcare infrastructure. As healthcare systems transition toward non-invasive and early detection methods, liquid biopsy has become a transformative solution for cancer screening and monitoring. Japan, being one of the most technologically advanced healthcare markets in Asia-Pacific, is rapidly integrating liquid biopsy technologies into clinical and research applications.
Liquid biopsy cancer screening is gaining traction due to its ability to detect cancer through blood samples, offering a less invasive and highly efficient alternative to traditional tissue biopsies. With Japan’s aging population and increasing healthcare expenditure, the demand for early-stage cancer detection tools is significantly rising. This article provides a comprehensive analysis of the Japan Liquid Biopsy Cancer Screening Market, covering market size, growth trends, key drivers, challenges, competitive landscape, and future opportunities.
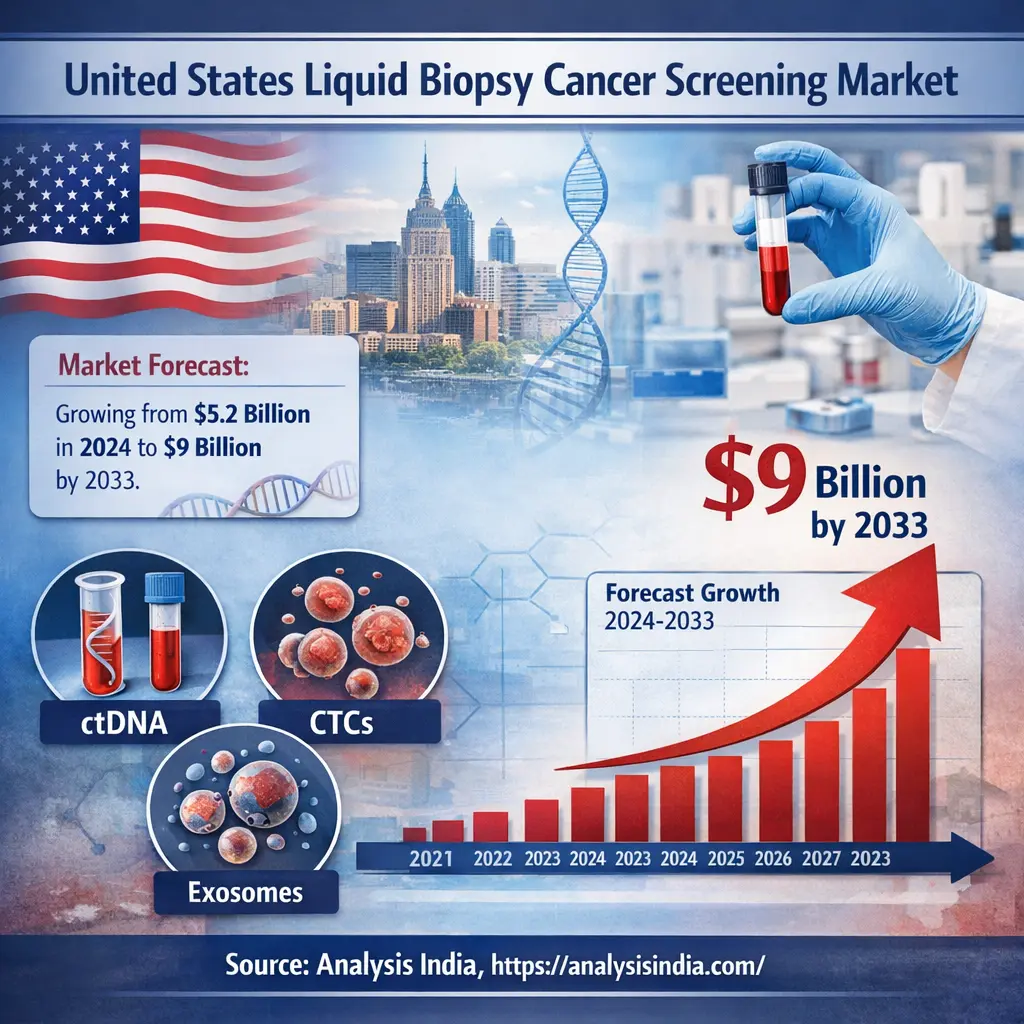
For a broader perspective, refer to the Global Liquid Biopsy Cancer Screening Market, which outlines worldwide trends shaping this industry.
Japan Liquid Biopsy Cancer Screening Market Size and Forecast
The Japan Liquid Biopsy Cancer Screening Market has shown consistent growth over recent years and is projected to expand steadily through 2033. According to market estimates, the market was valued at approximately USD 1.3 billion in 2024 and is expected to reach USD 2.4 billion by 2033, reflecting a gradual yet strong adoption curve.
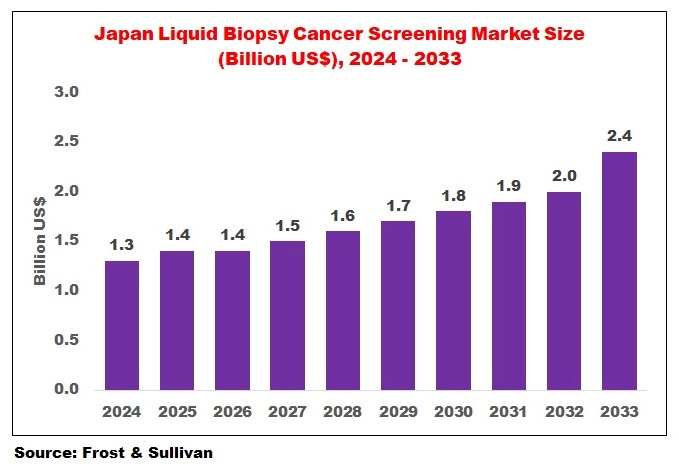
The steady increase highlights Japan’s commitment to adopting advanced diagnostic technologies. Compared to other developed markets such as the United States Liquid Biopsy Cancer Screening Market, Japan shows a more structured and regulated growth pattern, emphasizing quality and clinical validation.
Market Dynamics
Key Growth Drivers
The Japan Liquid Biopsy Cancer Screening Market is driven by several important factors that collectively enhance adoption and investment.
1. Rising Cancer Incidence in Japan
Japan faces a significant burden of cancer, particularly among its aging population. With increasing cases of lung, colorectal, breast, and gastric cancers, the need for early detection technologies has become more critical than ever. Liquid biopsy offers a viable solution by enabling early diagnosis, improving survival rates, and reducing treatment costs.
2. Aging Population and Healthcare Demand
Japan has one of the oldest populations globally. This demographic trend directly impacts the healthcare system, increasing demand for advanced diagnostic tools. The Japan Liquid Biopsy Cancer Screening Market benefits from this trend as elderly patients prefer minimally invasive diagnostic procedures.
3. Technological Advancements in Genomics
Japan is a leader in genomic research and precision medicine. Advanced sequencing technologies, such as next-generation sequencing (NGS), are widely adopted, enabling accurate detection of circulating tumor DNA (ctDNA). These advancements significantly enhance the reliability and effectiveness of liquid biopsy solutions.
4. Government Support and Healthcare Policies
The Japanese government actively supports cancer screening programs and research initiatives. Policies encouraging early detection and funding for innovative diagnostics are accelerating market growth. Public-private partnerships further strengthen the ecosystem.
For comparison with Asia’s fastest-growing market, explore the China Liquid Biopsy Cancer Screening Market, which shows rapid expansion driven by population scale.
Market Segmentation
By Biomarker Type
The Japan Liquid Biopsy Cancer Screening Market is segmented based on biomarker types, including:
- Circulating Tumor DNA (ctDNA)
- Circulating Tumor Cells (CTCs)
- Extracellular Vesicles
- Cell-free DNA (cfDNA)
Among these, ctDNA dominates due to its high sensitivity and specificity in detecting cancer mutations.
By Technology
- Next-Generation Sequencing (NGS)
- Polymerase Chain Reaction (PCR)
- Microarrays
- Others
NGS technology leads the market due to its ability to provide comprehensive genomic profiling.
By Cancer Type
- Lung Cancer
- Breast Cancer
- Colorectal Cancer
- Prostate Cancer
- Others
Lung cancer holds the largest share due to its high prevalence in Japan.
By End User
- Hospitals
- Diagnostic Laboratories
- Research Institutes
- Academic Centers
Hospitals dominate the segment due to increasing integration of liquid biopsy into clinical workflows.
Market Trends and Insights
Shift Toward Non-Invasive Diagnostics
The transition from traditional biopsy methods to liquid biopsy is a major trend in Japan. Patients and healthcare providers prefer non-invasive procedures that reduce discomfort and risk. This trend significantly boosts the Japan Liquid Biopsy Cancer Screening Market.
Integration with Artificial Intelligence
AI-driven analytics is enhancing the accuracy of liquid biopsy tests. Machine learning algorithms help identify patterns in genomic data, improving early detection rates.
Personalized Medicine Adoption
Japan is rapidly embracing personalized medicine. Liquid biopsy plays a crucial role in tailoring treatments based on individual genetic profiles, improving treatment outcomes.
Competitive Landscape
The Japan Liquid Biopsy Cancer Screening Market is highly competitive, with both global and domestic players actively participating. Key companies focus on innovation, partnerships, and regulatory approvals to strengthen their market presence.
Key Players
- Roche Diagnostics
- Thermo Fisher Scientific
- Guardant Health
- Illumina Inc.
- Qiagen N.V.
These companies are investing heavily in research and development to enhance product offerings and expand their footprint in Japan.
Challenges in the Market
Despite strong growth potential, the Japan Liquid Biopsy Cancer Screening Market faces several challenges.
High Cost of Testing
Liquid biopsy tests can be expensive, limiting accessibility for some patients. Although Japan’s healthcare system provides coverage, cost remains a barrier for widespread adoption.
Regulatory Complexity
Japan has stringent regulatory requirements for medical devices and diagnostics. While this ensures quality, it can slow down the introduction of new technologies.
Limited Awareness
Although awareness is increasing, many patients and healthcare providers are still unfamiliar with liquid biopsy technologies. Education and awareness campaigns are essential for market expansion.
Opportunities and Future Outlook
The future of the Japan Liquid Biopsy Cancer Screening Market looks promising, with several growth opportunities on the horizon.
Expansion of Screening Programs
Government-led screening programs are expected to incorporate liquid biopsy technologies, increasing adoption rates.
Collaboration with Research Institutions
Partnerships between companies and academic institutions will drive innovation and accelerate commercialization.
Growth in Precision Oncology
The rise of precision oncology will further boost demand for liquid biopsy solutions, as they play a key role in treatment selection and monitoring.
For a broader understanding of cancer diagnostics evolution, refer to the Cancer Screening Market Growth Trends Insights.
Japan in the Global Context
Japan holds a strategic position in the global market due to its technological capabilities and strong healthcare infrastructure. Compared to other regions:
- The United States Cancer Screening Market leads in innovation and commercialization.
- The China Cancer Screening Market excels in rapid adoption and scale.
- The Japan Cancer Screening Market focuses on quality and precision.
- The South East Asia Cancer Screening Market shows emerging growth potential.
Japan’s balanced approach ensures sustainable market development.
External Insights and Industry Validation
According to Frost & Sullivan, the adoption of liquid biopsy technologies in Japan is expected to accelerate due to advancements in genomic research and increasing demand for early cancer detection. Additionally, organizations such as the World Health Organization (WHO) emphasize the importance of early diagnosis in reducing cancer mortality rates globally.
Conclusion
The Japan Liquid Biopsy Cancer Screening Market is poised for steady growth, driven by technological advancements, demographic trends, and supportive healthcare policies. With a projected market size of USD 2.4 billion by 2033, Japan is set to become a key player in the global liquid biopsy landscape.
The market’s evolution reflects a broader shift toward non-invasive, personalized, and data-driven healthcare solutions. While challenges such as cost and regulatory complexity remain, ongoing innovation and government support are expected to overcome these barriers.
As the demand for early cancer detection continues to rise, the Japan Liquid Biopsy Cancer Screening Market will play a crucial role in transforming oncology diagnostics and improving patient outcomes. Strategic collaborations, increased awareness, and technological advancements will further strengthen its position in the coming years.
Frequently Asked Questions (FAQs)
What is the Japan Liquid Biopsy Cancer Screening Market size in 2024?
The market is valued at approximately USD 1.3 billion in 2024.
What is the projected market size by 2033?
The market is expected to reach USD 2.4 billion by 2033.
What drives the growth of this market?
Key drivers include rising cancer incidence, aging population, technological advancements, and government support.
Which technology dominates the market?
Next-generation sequencing (NGS) is the leading technology due to its high accuracy.
What are the major challenges?
High costs, regulatory complexity, and limited awareness are key challenges.