Introduction
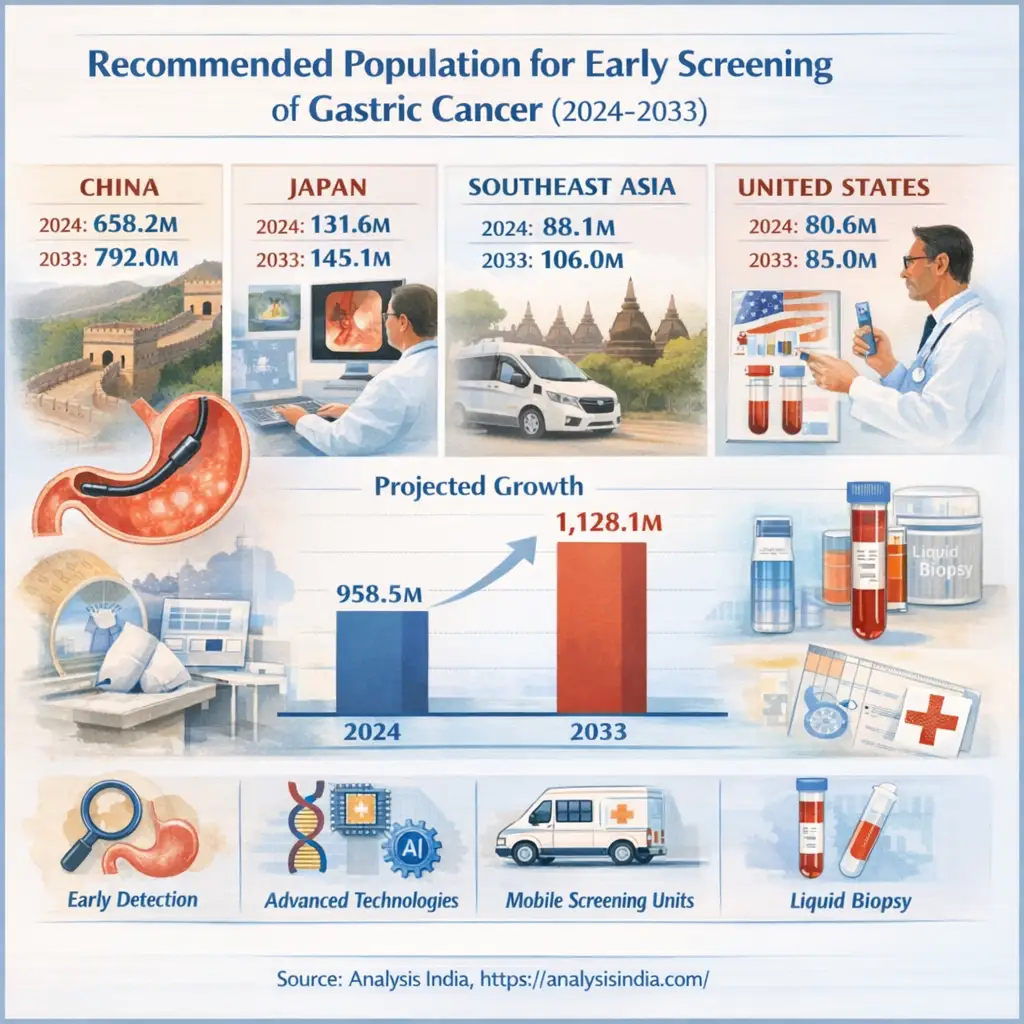
Early screening for gastric cancer has become a cornerstone of global cancer prevention strategies. The burden of gastric cancer remains high in Asia, with China, Japan, and Southeast Asia accounting for the majority of cases, while the United States continues to refine its targeted screening approach. Between 2024 and 2033, the recommended population for early screening of gastric cancer is projected to increase from 958.5 million to 1,128.1 million, reflecting growing awareness, policy support, and technological innovation.
This analysis builds upon our Global Gastric Cancer Screening Market pillar page and complements regional insights from China, Japan, United States, and South East Asia.
Understanding the Recommended Population for Early Screening of Gastric Cancer
The term recommended population for early screening of gastric cancer refers to individuals identified as high-risk based on factors such as age, genetic predisposition, dietary habits, Helicobacter pylori infection, and regional incidence rates.
From 2024 onward, healthcare authorities are increasingly refining screening guidelines to target:
- Individuals aged 40 and above in high-incidence regions
- Populations with a family history of gastric cancer
- Patients with chronic gastritis or H. pylori infection
- Communities with high consumption of smoked or salted foods
These criteria vary across regions, which explains the differences in screening population growth rates.
Global Screening Landscape
The global screening population is expanding steadily, driven by demographic shifts, healthcare reforms, and technological advancements. According to projections, the total recommended population for early screening of gastric cancer will grow at a compound annual growth rate (CAGR) of around 1.8% between 2024 and 2033. China leads this growth with a CAGR exceeding 2%, while Japan’s population remains stable due to demographic decline. Southeast Asia shows promising momentum, and the United States continues to adopt precision-based screening for high-risk groups.
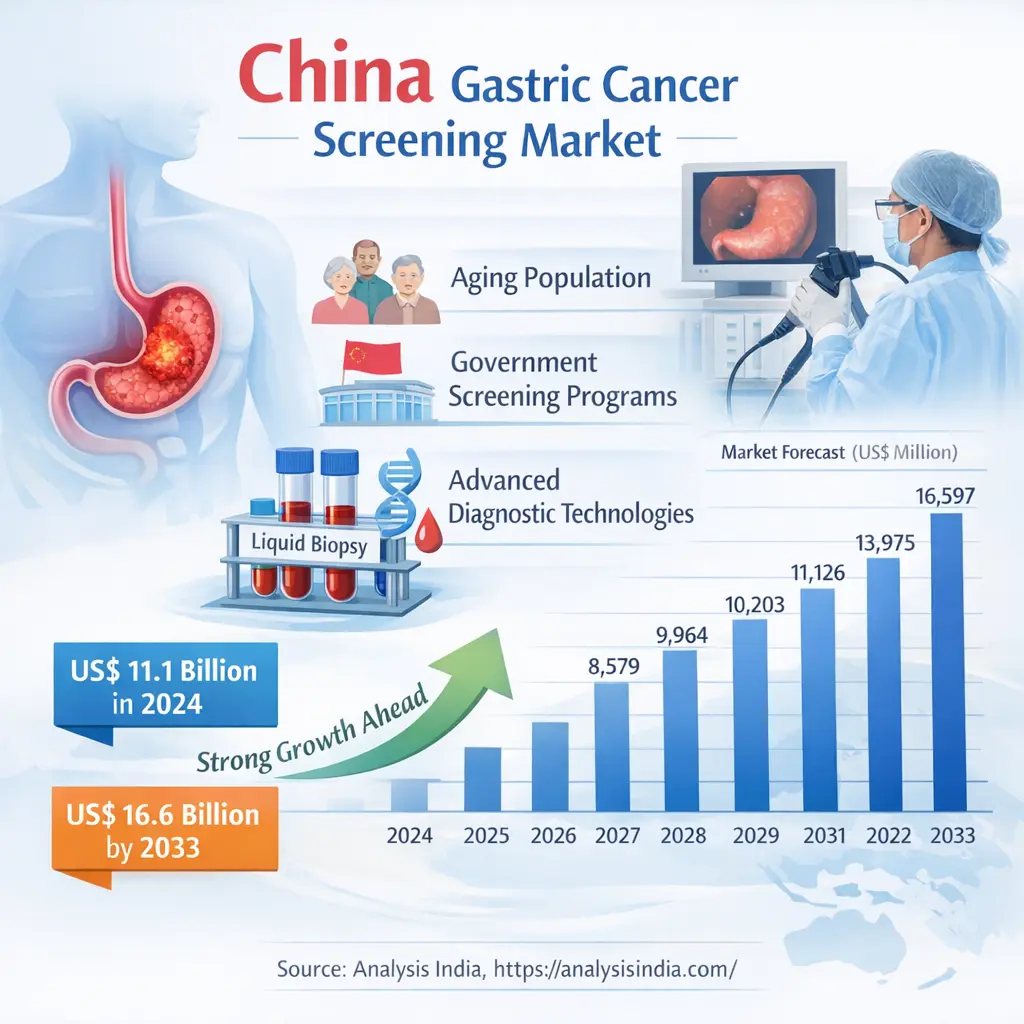
China Recommended Population for Early Screening of Gastric Cancer: Policy-Driven Expansion and Technological Leadership
China remains the epicenter of gastric cancer screening, with its recommended population for early screening of gastric cancer expected to rise from 658.2 million in 2024 to 792.0 million in 2033. This growth is underpinned by strong government initiatives and public health reforms. The National Health Commission of China continues to expand its Early Diagnosis and Treatment Program for Gastric Cancer, focusing on individuals aged 40 and above in high-incidence provinces such as Henan, Sichuan, and Shandong.
Policy frameworks emphasize endoscopic screening and Helicobacter pylori eradication, both proven to reduce gastric cancer mortality. The integration of AI-assisted endoscopy and capsule imaging has improved detection accuracy, enabling physicians to identify early-stage lesions with greater precision. Pilot programs in rural China have demonstrated that early detection can increase five-year survival rates to over 90%.
China’s healthcare infrastructure is also embracing cloud-based diagnostic systems that link regional hospitals with national cancer registries. This digital transformation supports real-time data sharing and enhances screening efficiency. As a result, China’s approach is not only expanding the recommended population but also setting a global benchmark for large-scale, technology-driven screening programs.
For deeper insights, explore our detailed regional analysis: China Gastric Cancer Screening Market 2033.
Japan Recommended Population for Early Screening of Gastric Cancer: Mature System with Technological Refinement
Japan’s recommended population for early screening of gastric cancer is projected to increase modestly from 131.6 million in 2024 to 145.1 million in 2033, reflecting a stable yet highly efficient system. The country’s National Cancer Center and Ministry of Health, Labour and Welfare have long maintained comprehensive screening programs targeting individuals aged 40 and above.
Japan’s success lies in its quality of screening rather than quantity. The country pioneered mass endoscopic screening decades ago and continues to refine its methods through AI-assisted image analysis, microRNA-based blood tests, and non-invasive biomarker panels. These innovations are reducing false negatives and improving early-stage detection.
Recent studies published in CiNii Journals and PubMed highlight that combining endoscopy with serum pepsinogen testing enhances diagnostic accuracy for early gastric lesions. Moreover, Japan’s healthcare system ensures equitable access through public insurance coverage, making screening affordable and routine.
Despite a declining population, Japan remains a global leader in screening precision, offering valuable lessons for emerging markets. For a detailed forecast, visit Japan Gastric Cancer Screening Market Forecast.
Southeast Asia Recommended Population for Early Screening of Gastric Cancer: Expanding Access and Awareness
Southeast Asia’s recommended population for early screening of gastric cancer is expected to grow from 88.1 million in 2024 to 106.0 million in 2033, marking one of the fastest-growing segments globally. The region’s progress is driven by WHO-backed initiatives and national health programs aimed at improving early detection.
Countries such as Thailand, Vietnam, and Indonesia are implementing community-based screening models that combine mobile diagnostic units with local health centers. These programs are particularly effective in rural areas where access to hospitals is limited. The WHO South-East Asia Regional Strategy for Cancer Prevention (2024–2030) emphasizes integrating gastric cancer screening into primary healthcare systems and promoting Helicobacter pylori eradication campaigns.
Technological adoption is accelerating, with low-cost endoscopy, AI-based triage tools, and portable imaging devices becoming more common. Case studies from Vietnam show that mobile screening units have doubled early detection rates in underserved communities.
Southeast Asia’s growth trajectory underscores the importance of public health education, international collaboration, and affordable technology in expanding screening coverage. For regional insights, explore South East Asia Gastric Cancer Screening Market.
United States Recommended Population for Early Screening of Gastric Cancer: Precision Medicine and Targeted Screening
The United States presents a different dynamic, with its recommended population for early screening of gastric cancer increasing from 80.6 million in 2024 to 85.0 million in 2033. Unlike Asia’s population-wide approach, the U.S. focuses on targeted screening for high-risk groups.
The American Gastroenterological Association (AGA) updated its guidelines in 2025, recommending screening for individuals with gastric intestinal metaplasia, family history of gastric cancer, or immigrant backgrounds from high-incidence regions. This precision-based strategy aligns with the broader trend toward personalized medicine.
Technological innovation is central to the U.S. approach. Liquid biopsy, AI-driven pathology, and non-invasive breath tests are increasingly used to detect early-stage gastric cancer. Research from Stanford Health Care and Johns Hopkins Medicine demonstrates that combining genetic risk profiling with non-invasive diagnostics can significantly improve detection rates.
The U.S. market also benefits from strong private-sector participation, with biotech firms developing advanced screening kits and digital platforms for remote monitoring. This integration of technology and data analytics ensures that screening remains efficient, targeted, and scalable.
For a comprehensive view, see United States Gastric Cancer Screening Market.
Recommended Population for Early Screening of Gastric Cancer for China, Japan, SEA and United States (2024–2033)
The following table provides a consolidated year-wise recommended population for early screening of gastric cancer in China, Japan, SEA and United States:
| Year | China | Japan | SEA | United States |
|---|---|---|---|---|
| 2024 | 944.5 | 48.1 | 142.7 | 102.6 |
| 2025 | 958.5 | 46.8 | 149.6 | 103.9 |
| 2026 | 976.3 | 46.6 | 152.5 | 105.7 |
| 2027 | 996.2 | 46.4 | 155.3 | 107.4 |
| 2028 | 1,015.8 | 46.3 | 160.7 | 108.8 |
| 2029 | 1,030.9 | 46.3 | 163.3 | 110.1 |
| 2030 | 1,045.5 | 46.2 | 165.8 | 111.2 |
| 2031 | 1,061.0 | 46.2 | 168.4 | 113.5 |
| 2032 | 1,079.4 | 46.1 | 171.1 | 114.8 |
| 2033 | 1,128.1 | 46.1 | 174.0+ | 116.2 |
Source: Frost & Sullivan, Analysis India
Global Technological and Policy Outlook
Across all regions, the next decade will witness a convergence of policy support and technological innovation. The World Health Organization (WHO) and International Agency for Research on Cancer (IARC) advocate for Helicobacter pylori screen-and-treat strategies as cost-effective interventions. Meanwhile, the rise of AI-assisted diagnostics, liquid biopsy, and mobile screening units is transforming accessibility and accuracy.
Countries investing in early detection infrastructure are seeing measurable declines in mortality. The integration of digital health platforms, cloud-based registries, and cross-border collaborations will further enhance global screening efficiency.
For related insights, explore our complementary pillars:
Role of Technology in Expanding Screening Population
Technological advancements are playing a crucial role in increasing the recommended population for early screening of gastric cancer.
Key Technologies
- AI-powered diagnostic tools
- Liquid biopsy
- Advanced imaging systems
- Telemedicine platforms
These innovations are making screening more accessible, affordable, and accurate, thereby expanding the eligible population.
Integration with Broader Cancer Screening Trends
The recommended population for early screening of gastric cancer is closely aligned with broader trends in the cancer screening ecosystem.
Key Industry Trends
- Shift toward preventive healthcare
- Increasing adoption of multi-cancer screening
- Rising investment in diagnostics
These trends are reinforcing the importance of early detection and expanding screening eligibility criteria.
Market Drivers Influencing Screening Population Growth
Several factors are driving the expansion of the recommended population for early screening of gastric cancer:
1. Rising Disease Burden
Gastric cancer remains prevalent in many regions, necessitating early detection.
2. Government Initiatives
Public health programs are increasing screening coverage.
3. Awareness Campaigns
Education is encouraging individuals to undergo screening.
4. Technological Advancements
Innovations are improving accessibility and accuracy.
Challenges in Expanding Screening Population
Despite growth, several challenges remain:
Limited Access in Rural Areas
Many regions still lack adequate healthcare infrastructure.
High Cost of Advanced Diagnostics
Expensive technologies limit widespread adoption.
Lack of Awareness
In some regions, awareness levels remain low.
Cultural Barriers
Social stigma can hinder participation in screening programs.
Recommended Population for Early Screening of Gastric Cancer Future Outlook (2024–2033)
The future of the recommended population for early screening of gastric cancer looks promising.
Expected Developments
- Increased adoption of AI-based screening
- Expansion of national screening programs
- Greater integration of digital health solutions
By 2033, the screening population is expected to reach new heights, driven by a global shift toward preventive healthcare.
Strategic Recommendations for Stakeholders
For Healthcare Providers
- Invest in early detection technologies
- Expand outreach programs
For Governments
- Implement nationwide screening initiatives
- Subsidize diagnostic procedures
For Investors
- Focus on emerging markets like Southeast Asia
- Support innovation in screening technologies
Conclusion
Between 2024 and 2033, the recommended population for early screening of gastric cancer will expand significantly, driven by China’s large-scale initiatives, Japan’s technological precision, Southeast Asia’s accessibility programs, and the United States’ targeted strategies.
This growth reflects a global commitment to early detection, policy innovation, and technological advancement. As countries continue to refine their screening frameworks, the focus will shift toward personalized prevention, data-driven diagnostics, and equitable access — ensuring that early screening becomes a universal standard in cancer care.
Frequently Asked Questions (FAQ)
What is the recommended population for early screening of gastric cancer?
The recommended population refers to individuals identified by health authorities as high-risk for gastric cancer, based on age, family history, and regional incidence rates. Between 2024 and 2033, this population is projected to grow globally from 958.5 million to 1,128.1 million.
Which region has the largest screening population?
China leads globally, with its recommended screening population expected to rise from 658.2 million in 2024 to 792.0 million in 2033, driven by government-backed screening programs and high incidence rates.
Why is Japan’s screening population stable compared to other regions?
Japan already has a mature nationwide screening program and a declining overall population, which results in slower growth. However, it remains a leader in adopting advanced technologies like AI-assisted endoscopy.
How is Southeast Asia addressing gastric cancer screening?
Southeast Asia is expanding screening programs through WHO-backed initiatives, low-cost endoscopy, and community-based outreach. The recommended population is expected to grow from 88.1 million in 2024 to 106.0 million in 2033.
What role does technology play in gastric cancer screening?
Technologies such as liquid biopsy, AI-driven diagnostics, and non-invasive biomarker tests are transforming screening approaches, especially in the United States and Japan, where precision medicine is emphasized.